| 动物的精子发生是指性腺中一小群叫做精原干细胞(SSCs)的细胞经过复杂的增殖、分化、减数分裂最终形成精子的过程。在胚胎发育早期,少数上胚层细胞首先形成原始生殖细胞,这些早期的生殖细胞经过增殖、迁徙和分化最终形成了SSCs。SSCs不仅能够经过分化变为能进行减数分裂的精母细胞,而且还能够重编程形成多能性干细胞。因此,SSCs是研究干细胞自我更新、分化、去分化、以及减数分裂的优秀细胞模型。精子发生的缺陷会导致男性不育和出生缺陷,而对其进行干预则可能发展出避孕、不育治疗、出生缺陷矫正、动物繁育、再生医学等方面的新技术。我们目前正在利用分子生物学、小鼠基因敲除、高通量组学和生物信息学等技术来研究SSCs增殖、分化、重编程以及利用多能性干细胞获得生殖细胞的分子机理。 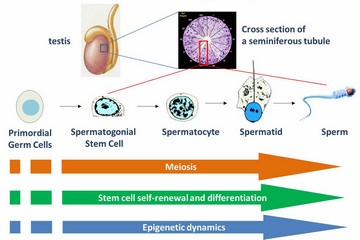
Mammalian spermatogenesis is an ideal model for fundamental biological processes such as meiosis, stem cell self-renewal and differentiation as well as epigenetic dynamics.
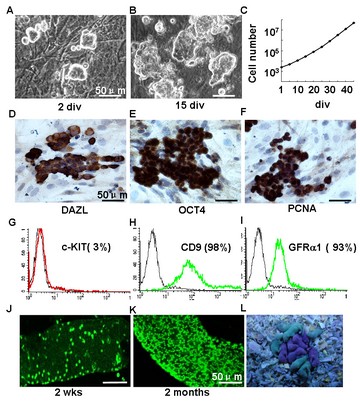
In vitro propagation of mouse spermatogonial stem cells (SSCs). SSCs express marker genes of stem cells and germ cells, and can be transfected by lentivirus. SSCs recover spermatogenesis after transplantation and give birth to transgenic mice.
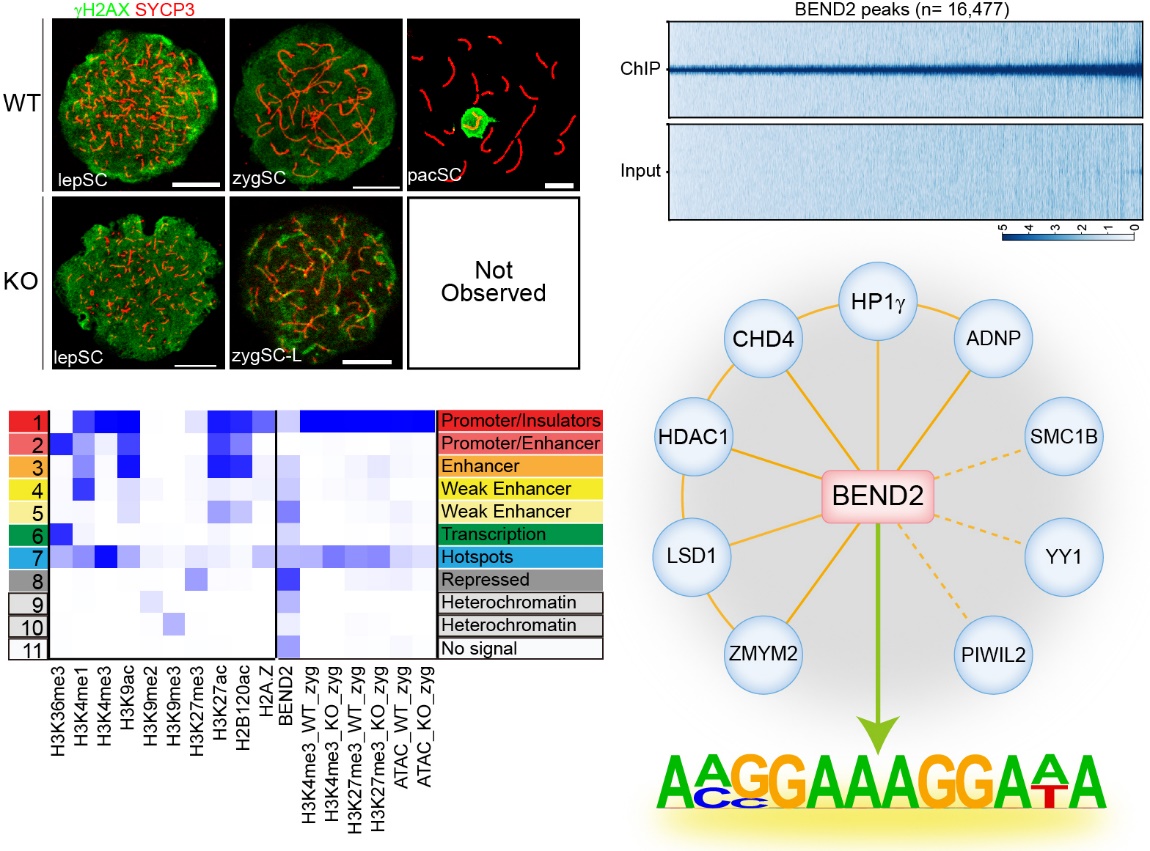
BEND2 is a previously unknown key regulator of meiosis, gene expression, and chromatin state during mouse spermatogenesis.
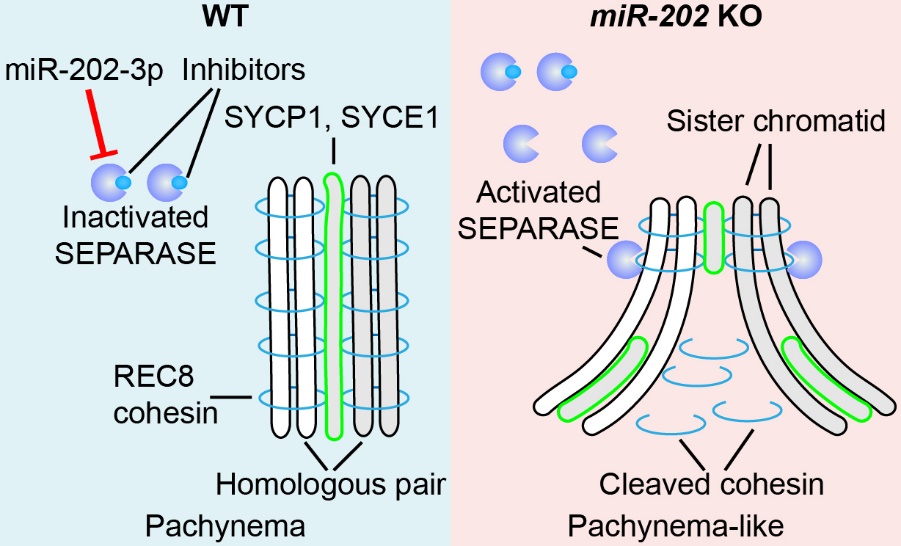
miR-202 safeguards meiotic progression by preventing premature SEPARASE-mediated REC8 cleavage during mouse spermatogenesis.
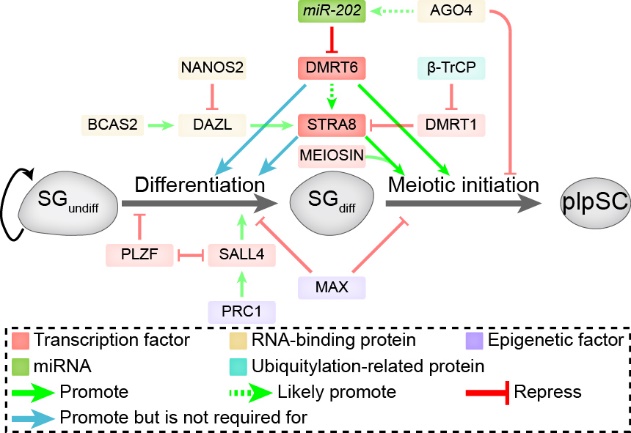
miR-202 prevents precocious spermatogonial differentiation and meiotic initiation. miR-202, DMRT6 and STRA8 act together as a module in the regulatory network of spermatogonial differentiation and meiotic initiation.
| 研究内容和目标:
1. 精原干细胞(SSCs)增殖、分化(减数分裂)以及去分化(重编程)研究。我们已经建立了精原干细胞的体外培养扩增以及多能性干细胞诱导形成生殖细胞的平台,为发展基于人工诱导生殖细胞的不育治疗技术奠定了基础。未来将面向男性不育治疗的应用目标,快速推进在体外获得“人造”精子的工作。
2. 精子发生的表观遗传调控机理。我们已经利用分离纯化的多种生精细胞结合组学技术,在DNA、RNA、蛋白质三个水平绘制哺乳动物精子发生基因调控的全局视图,利用基因敲除和分子生物学技术重点研究关键转录因子在精子发生中的功能,为男性不育的诊断提供了候选基因。未来我们将继续利用组学技术研究在生殖细胞形成和发育过程中基因表观遗传修饰的变化以及RNA和蛋白质的表达变化,发现对分化起关键作用的基因,并针对其作用机理设计出更加有效的体外分化诱导方案,为临床转化奠定基础。 代表性发表论文: - Wang X, Wang Y, Wang Y, Guo Y, Zong R, Hu S, Yue J, Yao J, Han C*, Guo J*, Zhao J*. Single-cell transcriptomic and cross-species comparison analyses reveal distinct molecular changes of porcine testes during puberty. Commun Biol. 2024 Nov 9;7(1):1478. doi: 10.1038/s42003-024-07163-9.
- Han C*. Gene expression programs in mammalian spermatogenesis. Development. 2024 Apr 15;151(8):dev202033. doi: 10.1242/dev.202033.
- Ning Y, Duo S, Lin X, Zhang H, Fei J, Zhang B, Zeng Y, Xie D, Chen J, Liu X, Han C*. Transcription factor PBX4 regulates limb development and haematopoiesis in mice. Cell Prolif. 2024 Jan 17:e13580. doi: 10.1111/cpr.13580. Epub ahead of print. PMID: 38230761.
- Li J, Lin X, Xie L, Zhao J, Han C*, Deng H*, Xu J*,A CRISPR/Cas9-based kinome screen identifies ErbB signaling as a new regulator of human naïve pluripotency and totipotency,Life Medicine, 2023, lnad037, https://doi.org/10.1093/lifemedi/lnad037
- Ma, L., Xie, D., Luo, M., Lin, X., Nie, H., Chen, J., Gao, C., Duo, S., and Han, C. (2022) Identification and characterization of BEND2 as a key regulator of meiosis during mouse spermatogenesis. Science Advances, 8(21):eabn1606.
- Chen, J., Gao, C., Luo, M., Zheng, C., Lin, X., Ning, Y., Ma, L., He, W., Xie, D., Liu, K., Hong, K., Han, C. (2022) MicroRNA-202 safeguards meiotic progression by preventing premature SEPARASE-mediated REC8 cleavage. EMBO Reports , 17;e54298.
- Chen, J., Gao, C., Lin, X., Ning, Y., He, W., Zheng, C., Zhang, D., Yan, L., Jiang, B., Zhao, Y., Hossen, MA., Han, C. (2021) The microRNA miR-202 prevents precocious spermatogonial differentiation and meiotic initiation during mouse spermatogenesis. Development, 148(24)
- Zheng ,C., Ouyang, Y., Jiang, B., Lin, X., Chen, J., Dong, M., Zhuang, X., Yuan, S., Sun, Q., Han, C. (2019) Non-canonical RNA polyadenylation polymerase FAM46C is essential for fastening sperm head and flagellum in mice. Biol Reprod, 100(6):1673-1
- Zhang, D., Xie, D., Lin, X., Ma, L., Chen, J., Wang, Y., Duo, S., Feng, Y., Zheng, C., Jiang, B., Ning, Y., Han, C.(2018) The transcription factor SOX30 is a key regulator of mouse spermiogenesis. Development,145. doi: 10.1242/dev.164723.
- Feng, Y., Ning, Y., Lin, X., Zhang, D., Liao, S., Zheng, C., Chen J., Wang Y., Ma L., Xie D., Han C.(2018) Reprogramming p53-Deficient Germline Stem Cells Into Pluripotent State by Nanog. Stem Cells Dev, 27:692-703.
- Hu, X., Shen, B., Liao, S., Ning, Y., Ma, L., Chen, J., Lin, X., Zhang, D., Li, Z., Zheng, C., Feng, Y., Huang, X., Han C.(2017) Gene knockout of Zmym3 in mice arrests spermatogenesis at meiotic metaphase with defects in spindle assembly checkpoint. Cell Death Di, 8:e2910.
- Chen, J., Cai, T., Zheng, C., Lin, X., Wang, G., Liao, S., Wang, X., Gan, H., Zhang, D., Hu, X., Wang, S., Li, Z., Feng, Y., Yang, F., and Han, C. (2017) MicroRNA-202 maintains spermatogonial stem cells by inhibiting cell cycle regulators and RNA binding proteins, Nucleic Acids Res. 45(7):4142-4157.
- Tu, Z., Bayazit, M. B., Liu, H., Zhang, J., Busayavalasa, K., Risal, S., Shao, J., Satyanarayana, A., Coppola, V., Tessarollo, L., Singh, M., Zheng, C., Han, C., Chen, Z., Kaldis, P., Gustafsson, J. A., and Liu, K. (2017) Speedy A-Cdk2 binding mediates initial telomere-nuclear envelope attachment during meiotic prophase I independent of Cdk2 activation, Proc Natl Acad Sci U S A, 114:592-597
- Chen, M., Zhang, L., Cui, X., Lin, X., Li, Y., Wang, Y., Wang, Y., Qin, Y., Chen, D., Han, C., Zhou, B., Huff, V., and Gao, F. (2017) Wt1 directs the lineage specification of sertoli and granulosa cells by repressing Sf1 expression, Development 144, 44-53
- Wang, S., Wang, X., Ma, L., Lin, X., Zhang, D., Li, Z., Wu, Y., Zheng, C., Feng, X., Liao, S., Feng, Y., Chen, J., Hu, X., Wang, M., and Han, C. (2016) Retinoic Acid Is Sufficient for the In Vitro Induction of Mouse Spermatocytes, Stem cell reports 7, 80-94.
- Lin, X., Han, M., Cheng, L., Chen, J., Zhang, Z., Shen, T., Wang, M., Wen, B., Ni, T., and Han, C. (2016) Expression dynamics, relationships, and transcriptional regulations of diverse transcripts in mouse spermatogenic cells, RNA biology 13, 1011-1024.
- Wu, Y., Hu, X., Li, Z., Wang, M., Li, S., Wang, X., Lin, X., Liao, S., Zhang, Z., Feng, X., Wang, S., Cui, X., Wang, Y., Gao, F., Hess, R. A., and Han, C. (2016) Transcription Factor RFX2 Is a Key Regulator of Mouse Spermiogenesis, Scientific reports 6, 20435.
- Yang, Y., Feng, Y., Feng, X., Liao, S., Wang, X., Gan, H., Wang, L., Lin, X., and Han, C. (2016) BMP4 Cooperates with Retinoic Acid to Induce the Expression of Differentiation Markers in Cultured Mouse Spermatogonia, Stem cells international 2016, 9536192.
- Xue, Y., Lameijer, E. W., Ye, K., Zhang, K., Chang, S., Wang, X., Wu, J., Gao, G., Zhao, F., Li, J., Han, C., Xu, S., Xiao, J., Yang, X., Ying, X., Zhang, X., Chen, W. H., Liu, Y., Zhang, Z., Huang, K., and Yu, J. (2016) Precision Medicine: What Challenges Are We Facing?, Genomics, proteomics & bioinformatics 14, 253-261.
- Gao, Y., Bai, X., Zhang, D., Han, C., Yuan, J., Liu, W., Cao, X., Chen, Z., Shangguan, F., Zhu, Z., Gao, F., and Qin, Y. (2016) Mammalian elongation factor 4 regulates mitochondrial translation essential for spermatogenesis, Nature structural & molecular biology 23, 441-449.
- #Zhu, L., #Han, C. S., Cao, Z. L., Wang, Z. B., Han, R. G., Wang, B., and Sun, Q. Y. (2015) Confocal Microscopic Analysis of the Spindle and Chromosome Configurations of in vitro-Matured Oocytes from Different Types of Polycystic Ovary Syndrome Patients, Gynecologic and obstetric investigation 80, 179-186.
- Wang S.,Wang Xx.,Wu Yj., and *Han, C. (2015) ,IGF-1R Signaling Is Essential for the Proliferation of Cultured Mouse Spermatogonial Stem Cells by Promoting the G2/M Progression of the Cell Cycle,Stem Cells and Development 24,471-483。
- Li, Y., Wang, X., Feng, X., Liao, S., Zhang, D., Cui, X., Gao, F., and *Han, C. (2014) Generation of male germ cells from mouse induced pluripotent stem cells in vitro, Stem Cell Res 12, 517-530.
- *Han, C. (2014) ,Which one is the real matchmaker for the pair?,Asian Journal of Andrology 16,667-668。
- Gan, H., Wen, L., Liao, S., Lin, X., Ma, T., Liu, J., Song, C.-x., Wang, M., He, C., *Han, C., and *Tang, F. (2013) Dynamics of 5-hydroxymethylcytosine during mouse spermatogenesis,Nature Communications 4, 1995.
- Wang, F., Yang, Y., Lin, X., Wang, J. Q., Wu, Y. S., Xie, W., Wang, D., Zhu, S., Liao, Y. Q., Sun, Q., Yang, Y. G., *Guo, C., *Han, C., and *Tang, T. S. (2013) Genome-wide Loss of 5-hmC is a Novel Epigenetic Feature of Huntington's Disease, Human Molecular Genetics 22, 3641-3653.
- Wu, Y., Liao, S., Wang, X., Wang, S., Wang, M., and *Han, C. (2013) HSF2BP represses BNC1 transcriptional activity by sequestering BNC1 to the cytoplasm, FEBS Letters 587, 2099-2104.
- hi, Y., Zhuang, X., Xu, B., Hua, J., Liao, S., Shi, Q., *Cooke, H. J., *Han, C. (2013) SYCP3-like X-linked 2 is expressed in meiotic germ cells and interacts with synaptonemal complex central element protein 2 and histone acetyltransferase TIP60, Gene 527, 352-359.
- Gan, H., Cai, T., Lin, X., Wu, Y., Wang, X., *Yang, F., and *Han, C. (2013) Integrative Proteomic and Transcriptomic Analyses Reveal Multiple Post-transcriptional Regulatory Mechanisms of Mouse Spermatogenesis, Mol Cell Proteomics 12, 1144-1157.
- Hou, X., Zhang, W., Xiao, Z., Gan, H., Lin, X., Liao, S., and *Han, C. (2012) Mining and characterization of ubiquitin E3 ligases expressed in the mouse testis, BMC Genomics 13, 495.
- Zhang, Y., Wang, S., Wang, X., Liao, S., Wu, Y., and *Han, C. (2012) Endogenously produced FGF2 is essential for the survival and proliferation of cultured mouse spermatogonial stem cells, Cell Research 22, 773-776.
- Gan, H., Lin, X., Zhang, Z., Zhang, W., Liao, S., Wang, L., and *Han, C. (2011) piRNA profiling during specific stages of mouse spermatogenesis, RNA 17, 1191-1203.

|

